Targeting Hallmarks of Aging Across Disease
Loss of repair capacity, chronic inflammation, mitochondrial dysfunction and genomic instability are shared hallmarks of aging and age-associated disease. We apply chemical switching to disease-relevant models, including patient-derived systems, to restore biological resilience rather than suppress signalling. Further, we develop the tools to selectively reprogram, deliver or execute function across tissues and organs. Our goal is to establish enzyme activation of DNA repair protein through ORCAs and COPs as a general strategy to intervene across cancer, fibrotic, metabolic and neurodegenerative diseases.
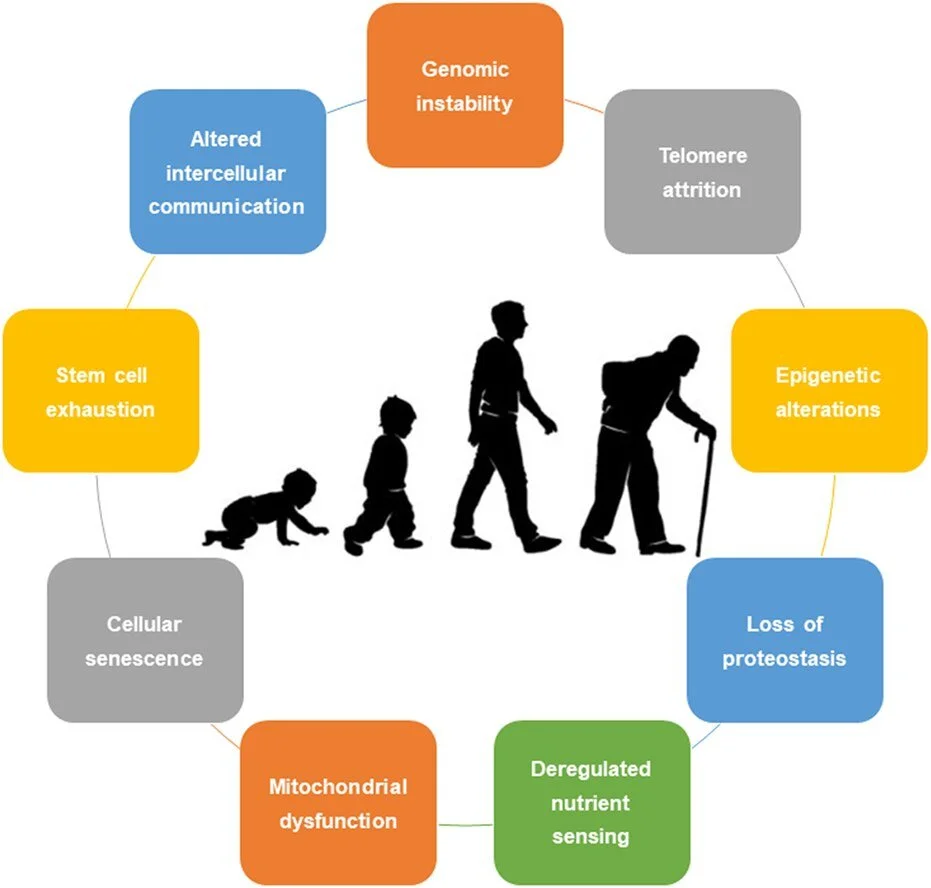
DNA Repair and the Hallmarks of Aging: Aging is characterized by a progressive loss of DNA repair capacity, leading to genomic instability, chronic inflammation, mitochondrial dysfunction, and altered cellular fate. Defects in DNA repair enzymes amplify multiple hallmarks of aging across tissues, linking genome maintenance directly to degenerative disease, cancer, and loss of biological resilience. By understanding and restoring DNA repair enzymology, we aim to intervene upstream of diverse age-associated pathologies rather than targeting their downstream consequences.
Wikimedia: Rebelo-Marques, De Sousa Lages, Andrade, Ribeiro, Mota-Pinto, Carrilho and Espregueira-Mendes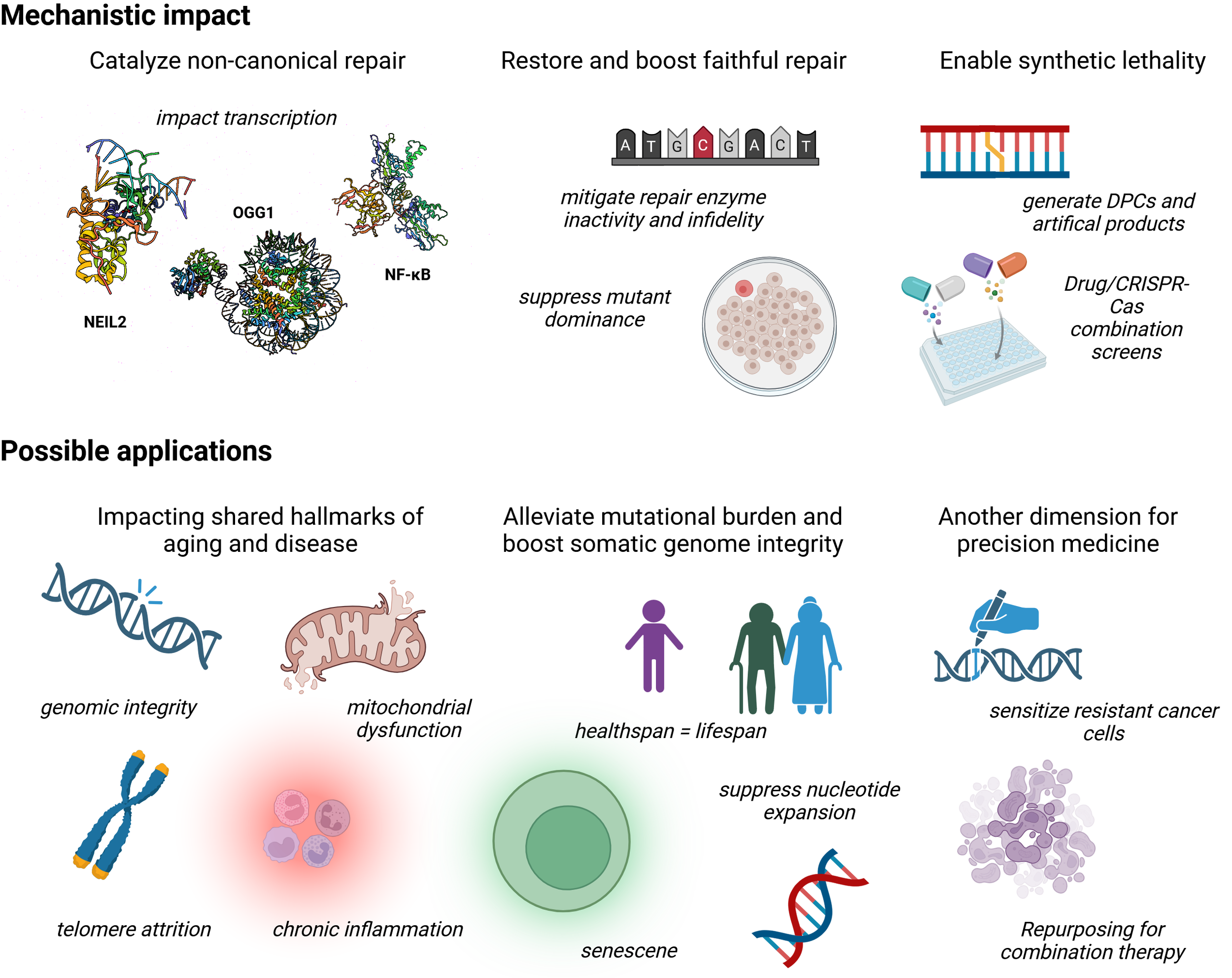
Chemical switching of DNA repair offers significant mechanistic impact and enables a range of possible applications: Mechanistic impact – In catalyzing biochemical reactions not commonly performed by the enzyme of interest, residence time, product and interaction partners may be affected. Both NEIL2 (PDB: 8TH9) and OGG1 (PDB: 8VX6) have been shown to affect transcription, albeit by opposed mechanisms. Here, especially OGG1 has been shown to recruit NF-ĸB (PDB: 7VUP) affecting release of proinflammatory cytokines. A disruption of this process leads to significant rewriting of inflammatory signaling; mutation, lower protein levels and reduced post-translational modification negatively affect OGG1 function. An improvement may restore repair processes and suppress mutation rates; The catalysis of new reaction pathways allows for novel DNA repair products which could enable synthetic lethality. DPC and non-canonical products shape new pathways within DNA repair, distinct from naïve conditions and entirely uncharacterized; Possible applications – Dual function medicines would address hallmarks of aging and disease at the same time. In DNA repair the immediate solutions would impact genomic integrity, mitochondrial dysfunction, telomere attrition, chronic inflammation and cellular senescence. An alleviated burden of mutation and with it a boosted somatic genomic integrity would thus contribute to an equal health- and lifespan. One of the underlying mechanisms identified is increased nucleotide expansion in differentiated tissue. Another dimension in precision medicine enabled by pathway rewriting can lead to sensitization of prior resistant cancer cells. Through full characterization of the rewritten pathway, entirely novel combination therapies of previously ineffective treatments are possible.
