Chemical Switching
Chemical switching is a technology to expand and reprogram enzyme function rather than inhibit it (concept in Visnes et al. ChemBioChem2025). We develop organocatalytic switches (ORCAs) and change-of-function protein variants (COPs) that participate directly in enzymatic reactions, enhancing activity or enabling new catalytic outcomes in living systems. Focusing on targets that are implicated in the Hallmarks of aging and Hallmarks of Cancer, we take a technology-centric approach to DNA repair. This activation-based strategy allows us to rewrite repair pathways, modulate inflammatory signalling and explore synthetic lethality through catalytic rewiring of human physiology.
The Essentials of Our Approach
Human physiology is limited by evolutionary constraints that are increasingly evident in the context of aging, processed diets and modern environmental stressors. While traditional medicine has largely operated under an inhibitor paradigm, which focuses on blocking or degrading protein functions, there is a significant lack of research into enzymatic activation or the enhancement of canonical functions. A transformative solution lies in the concept of chemical switching, which aims to move beyond inhibition by mapping and exploiting the dark activities of the human proteome. These dark activities represent alternative, often unmapped reaction pathways that enzymes are chemically capable of performing but do not utilize as their primary function in living cells.
The practical application of this technology involves manipulating biochemical transformations to lower the activation energy of formerly unfavoured reactions. This is achieved through two primary methods: Organocatalytic Switches (ORCAs) and change-of-function Protein Variants (COPs). ORCAs are small molecules that bind to an enzyme's active site and participate directly in the chemistry, for example, by facilitating proton abstraction to trigger elimination reactions. COPs on the other hand, involve engineering proteins with covalent warheads or amino acid mutations to introduce new chemical properties directly into the protein structure, creating hyperactive enzymatic constructs.
Listen to the concept Podcast
Want to know more?
A Video Summary
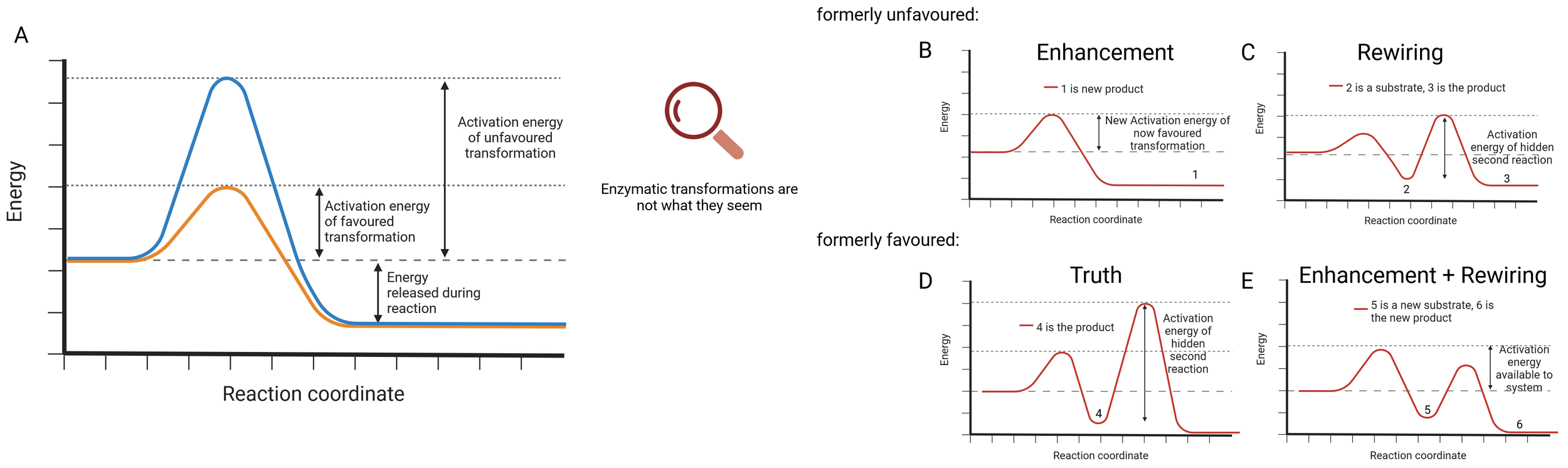
“Dark activities” of enzymes and virtually all proteins are underlying favoured as well as unfavoured chemical reactions: A) Activation energies define which enzymatic reactions proceed. Redefining what we treat as enzymatic function, activity and transformation opens the possibility to chemically switch all levels of enzyme control; B) Formerly unfavoured chemical reactions are improved to generate new products, which equals the enhancement of rudimentary or slow reactions; C) The reaction profile may also underlie secondary transformations in which the reaction intermediate may be an additional substrate. Both original substrate and former intermediate may be transformed into an entirely new product, effectively rewiring the reaction pathway; D) A favoured biochemical reaction may indeed only be the first step of a potential and hidden enzymatic cascade; E) Lowering the activation barrier of the following steps successfully rewires the enzymatic cascade by enhancing one transformation.
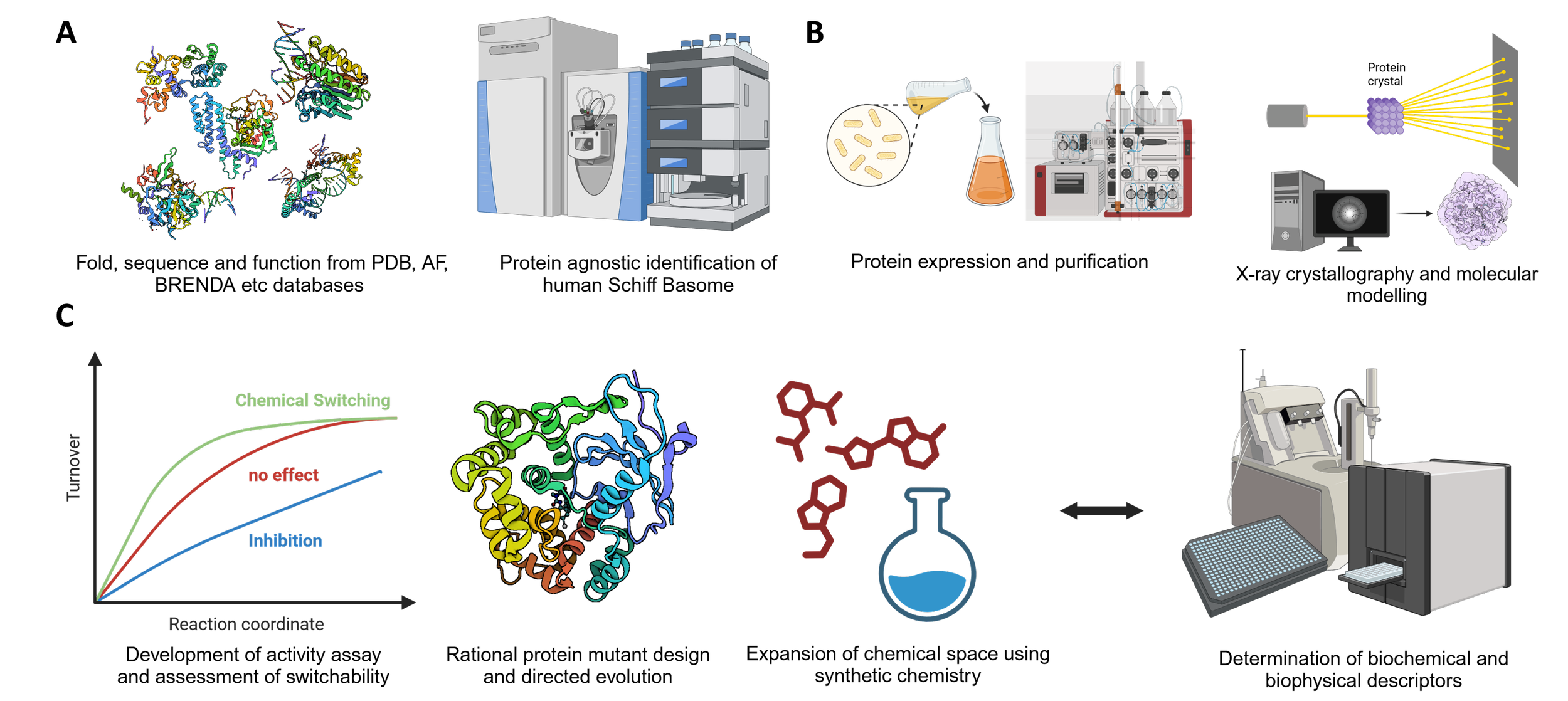
Identified challenges to broadly establish chemical switching: A) Data-driven phase: An open-access database is required that combines proteomewide fold, sequences, and functions sourced from PDB, AlphaFold, and BRENDA databases. A manageable and actionable subset needs to be informed by the elucidated human Schiff Basome. Here, agnostic mass spectrometry proteomics will yield the relevant players using a breadth of exogeneous and endogenous stress sources. B) Target enabling phase: In the next step, all identified members need to be purified or sourced from the community. Structural biology and molecular modeling approaches will increase insight into the molecular mechanism informing back into the initial database. C) Traction phase: All candidate proteins are assessed for productive Schiff base cleavage in specific biochemical assays. Information from the assembled model informs potential gain-of-function proteins that compete with canonical function. Chemical space of organocatalytic switches is discovered by medicinal chemistry efforts that include complex optimization restrictions. All potential proteins and small molecules need to be thoroughly investigated using biochemical and biophysical assays
